
IVF NewsWebinar: ARE ONLINE WEBINARS HELPING EMBRYOLOGISTS TO PASS THE CERTIFICATION EXAMS ?
Marc Van den Bergh 26 April 2024
Based on a published curriculum, 39 webinars were and are still accessible as records to view on demand between begin 2022 and september 2023. [ Full Article ] News: ART & Embryology training program
Chennai Fertility Center and Research Institute 23 April 2024

Training Batch Schedule 2024
The International School of Embryology a unit of Chennai Fertility Centre and Research Institute was established to offer training in Advanced Reproductive Techniques and Embryology for clinicians and embryologists. It will help them to know in-depth knowledge and have good hands-on training. The members of our teaching faculty aim to bring Clinician and Embryologists to the highest level of knowledge about Assisted Reproductive Technology and practical capability. Our courses cover basics in Andrology, embryology, ICSI, and cryosciences (Hands-on). Limited Seats. For admission Contact 9003111598 / 8428278218 [ Full Article ] Join Our IVF Newsletter for Updates & Latest JobsNews: MERLN Pioneers New Approach for Studying Monozygotic Twin Formation
IVF.net Newsdesk 21 April 2024
Researchers at Maastricht UMC+ and the MERLN Institute have developed the first embryo structure capable of mimicking the formation of human identical twins, utilizing only stem cells without the need for gametes. This breakthrough allows unprecedented insight into the formation of identical twins, where the rapid expansion of the blastocyst—a sac comprised of placental cells that houses the stem cells from which life originates—causes the embryo to split into two. Published in the prestigious journal Advanced Materials, this research leverages a sophisticated technological platform that has been revealing intricate biological processes over the years, facilitating the controlled growth of cells, tissues, organs, and embryos. The focus of this current study is on understanding the crucial early stages of healthy embryo implantation and development, offering a new avenue to explore foundational micro-processes that have traditionally been obscured within the womb. This research aims to enhance biomedical care, making it more accessible and affordable globally. Applications in Medicine The creation of synthetic embryos from stem cells has reached a fidelity that provides essential insights into natural embryonic development. This research has practical implications, particularly in enhancing our understanding of miscarriages and infertility. It offers potential solutions to fertility issues and improvements in contraception methods. Historically, three-quarters of identical twins have shared a single placenta, a phenomenon that until now was poorly understood. This new discovery could illuminate the underlying mechanisms of this occurrence. Moreover, twin pregnancies often involve complications that manifest during the early stages of implantation, which can now be studied more effectively, potentially leading to preventive measures or treatments. Innovative Research Platforms: High Throughput and Implantation-on-Chip In the lab, these early-stage synthetic embryos are cultivated for up to 14 days and subjected to thousands of parallel high-throughput experiments. Each experiment uses unique combinations of growth factors and signaling molecules to determine the optimal conditions for embryonic growth during the first week, including precise timing. For the second week of growth, researchers have developed an "implantation-on-chip" platform, which enables the cultivation and examination of small samples of uterine tissue on a microfluidic chip to optimize conditions for embryo implantation in the uterus. Lead researcher Erik Vrij noted that this platform could predict the success of treatments following procedures like IVF and PGT. Enhancing Medical Care Through Advanced Technologies The integration of robotics and machine learning is refining the simulation of biological processes, with the high-throughput approach increasing the likelihood of successfully creating a twin embryo model. According to MERLN's founder, Professor Clemens van Blitterswijk, the identified formulas from this research could lead to the development of tissue-specific stem cells, tissues, and organ parts for patient treatment. The goal is to enable these advancements on a scale that could significantly help a large number of people while keeping costs manageable. This research forms part of the PhD thesis of Dorian Luijkx under the supervision of Erik Vrij, Stefan Giselbrecht, Clemens van Blitterswijk, with collaboration from Asli Ak and Ge Guo of the University of Exeter. Source: MERLN Develops the First Model for Monozygotic Twinning [ Full Article ] Announcement: Cryopreservation in Assisted Reproduction - A Practitioner's Guide to Methods, Management, and Organization
IVF.net Newsdesk 21 April 2024

We are excited to announce the upcoming release of "Cryopreservation in Assisted Reproduction: A Practitioner's Guide to Methods, Management, and Organization" by Springer Nature. This comprehensive guide delves deep into the critical aspects of cryopreservation, a cornerstone of assisted reproductive technologies, and goes beyond traditional protocol books by covering organizational strategies, auditing, and inventory management. Highlights Include:
Innovative Breakthroughs in Reproductive Technology:
Featuring contributions from over 50 key opinion leaders in the field of in vitro fertilization (IVF), this book is a treasure trove of knowledge and expertise. It serves as an essential resource for clinicians, embryologists, lab technologists, IVF lab directors, and researchers. The guide presents well-established procedures in cryopreservation while also highlighting novel techniques and the latest innovations in the field. Your Comprehensive Guide to Cryobiology: This publication promises to revolutionize the approach to cryopreservation in assisted reproduction, focusing on both the scientific and practical aspects of managing and organizing advanced reproductive technologies. Prepare to dive into a world of groundbreaking innovations and advanced platforms that are setting new standards in the field of reproductive medicine. "Cryopreservation in Assisted Reproduction" is set to be an indispensable resource, guiding professionals through the complexities of modern reproductive technologies with the latest scientific advancements. https://link.springer.com/book/9783031582134 [ Full Article ] Conference: IVIRMA Assisted Reproduction Symposium
IVIRMA Global 02 April 2024

We are thrilled to extend a warm invitation to you for the upcoming 2nd IVIRMA Assisted Reproduction Symposium scheduled from May 8th to 11th, 2024. This four-day symposium is specially designed for physicians, embryologists, nurses, and geneticists. It will be conducted online and is free of charge. The event will feature distinguished international speakers who will cover cutting-edge topics such as PGT-A findings, experimental therapies for ovarian aging, non-invasive embryo assessment, immunological treatments, and the significance of the endometrium in reproductive success. Chairman
Scientific Secretary
Scientific Committee Members
[ Full Article ] Webinar: UP CLOSE AND CLINICAL
International IVF Initiative 02 April 2024

3pm ET/ 8pm UK/ 9pm CET, Tuesday 23rd April 2024. Moderators: Panelist: Dr. Sergio Papier Presentations: Moonshots and last miles: what it may take to treat Opening the black box: why do euploid blastocysts fail to An artificial intelligence tool predicts blastocyst Storage trends, usage and disposition outcomes following egg Q and A [ Full Article ] News: Unveiling the Mysteries of Egg Fertilization: The Role of ZP2 Cleavage and Egg Coat Hardening
IVF.net Newsdesk 02 April 2024
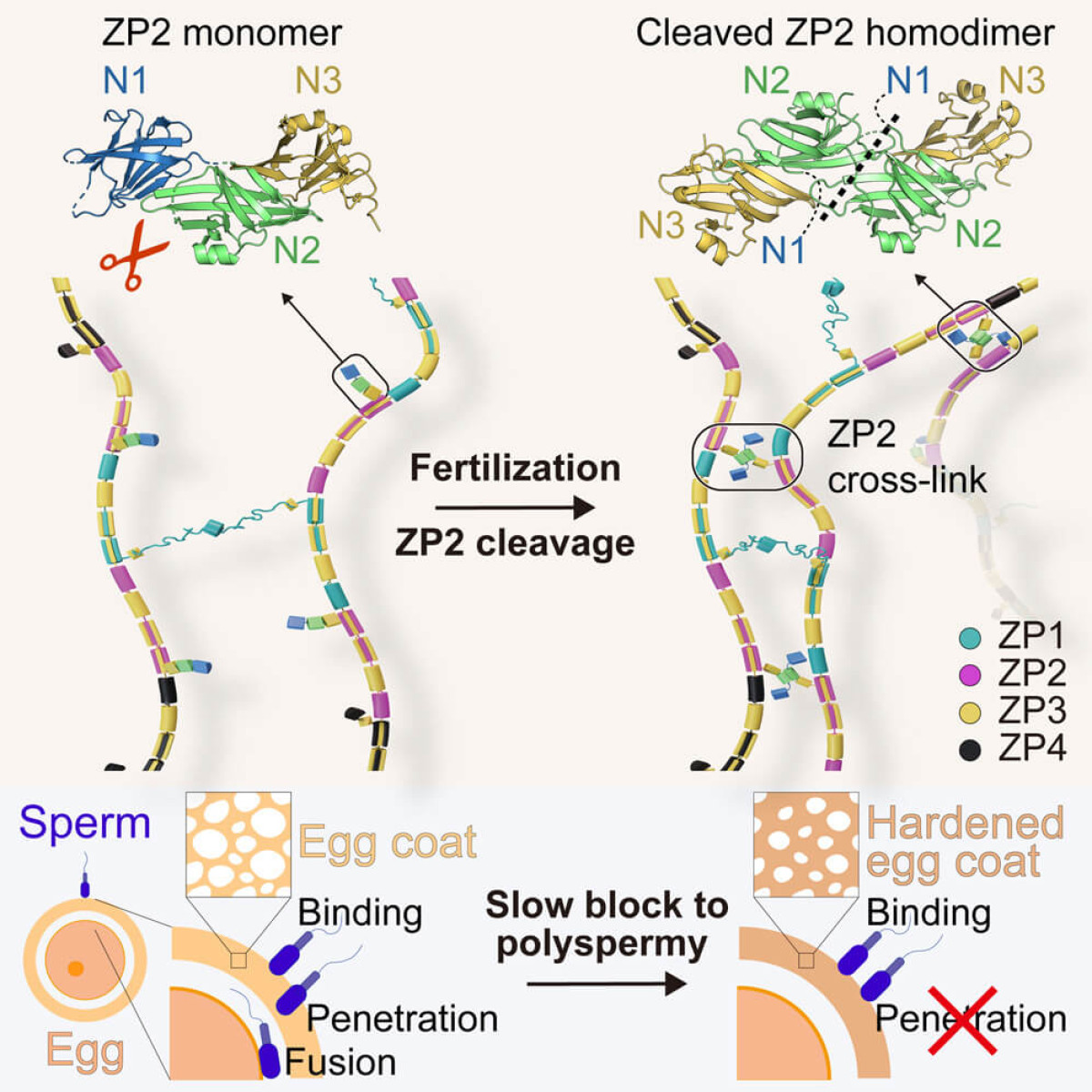
Fertilization, the pivotal moment when a single sperm merges with an egg to form a new life, is a marvel of biological precision. Beyond the romance of conception lies a complex ballet of molecular interactions, finely tuned to ensure that only one sperm can succeed. Central to this process is the zona pellucida (ZP), the protective egg coat that undergoes remarkable changes immediately after fertilization to prevent any other sperm from entering the egg. Recent groundbreaking research has shed light on the molecular intricacies of this process, particularly focusing on the role of the ZP2 glycoprotein, a key player in the hardening of the egg coat that blocks polyspermy – the penetration of an egg by more than one sperm. The Crucial Role of ZP2 in FertilizationThe ZP, a glycoprotein envelope surrounding the egg, is the first line of interaction between the egg and the sperm. It plays a critical role in species-specific sperm recognition and acts as a gatekeeper, ensuring that only one sperm can fertilize the egg. Following the initial fertilization event, the ZP undergoes a transformation, hardening to prevent any subsequent sperm from penetrating the egg. The mechanism behind this transformation has long been a subject of fascination and study within the scientific community. Recent studies have uncovered a critical piece of this puzzle: the cleavage of ZP2, a major component of the ZP structure. Before fertilization, ZP2 is primed to interact with sperm. However, once fertilization occurs, ZP2 is cleaved, undergoing a significant transformation that leads to the hardening of the ZP. From Cleavage to Hardening: The Journey of ZP2The journey from ZP2 cleavage to egg coat hardening is a complex one, involving the oligomerization of the cleaved ZP2. Oligomerization is a process by which proteins bind together, forming a structure that is significantly different from that of the individual proteins. In the case of ZP2, cleavage triggers this oligomerization, leading to extensive cross-linking between ZP filaments, the structural components of the ZP. This cross-linking effectively reinforces the ZP, making it impenetrable to additional sperm. This revelation came from both biochemical and structural studies, including the analysis of vertebrate egg coat filaments and computational models of human ZP. These studies have provided unprecedented insights into the molecular architecture of the egg coat and the dramatic alterations it undergoes following fertilization. Implications and Future DirectionsThe findings from these studies are not just academically intriguing; they have practical implications for understanding human fertility and developing treatments for fertility issues. By illuminating the molecular basis of ZP hardening, this research opens the door to potential interventions for conditions related to polyspermy, which can lead to fertilization failures and early embryonic loss. Moreover, the detailed understanding of ZP2's role in fertilization could lead to novel contraceptive methods that mimic the natural blocking mechanisms of the egg coat. On the flip side, for couples facing infertility challenges, tweaking the process of ZP2 cleavage and oligomerization could enhance the chances of successful fertilization during assisted reproductive techniques. Concluding ThoughtsThe dance of fertilization, with its tightly regulated steps and precise molecular cues, is a testament to the complexity of life's beginnings. The recent advances in our understanding of ZP2 cleavage and egg coat hardening add a fascinating chapter to the story of human reproduction. As we continue to unravel the mysteries of fertilization, we not only deepen our appreciation for the wonders of biology but also arm ourselves with the knowledge to tackle some of the most pressing challenges in reproductive health. The journey from a single sperm's successful penetration of the egg to the hardening of the egg coat is a marvel of biological engineering, ensuring the uniqueness of each fertilization event. In the grand scheme of life, it's a reminder of the delicate balance and intricate processes that underpin our very existence. Source Cell - ZP2 cleavage blocks polyspermy by modulating the architecture of the egg coat [ Full Article ] News: ART & Embryology training program
Chennai Fertility Centre and Research Institute 01 April 2024

Training Batch Schedule 2024 May = 06th to 20th 2024 The International School of Embryology a unit of Chennai Fertility Centre and Research Institute was established to offer training in Advanced Reproductive Techniques and Embryology for clinicians and embryologists. It will help them to know in-depth knowledge and have good hands-on training. The members of our teaching faculty aim to bring Clinician and Embryologists to the highest level of knowledge about Assisted Reproductive Technology and practical capability. Our courses cover basics in Andrology, embryology, ICSI, and cryosciences (Hands-on). Limited Seats. For admission Contact 9003111598 / 8428278218 [ Full Article ] Announcement: Teaser Drops for Netflix Show "JOY" - The IVF Pioneers Story
IVF.net Newsdesk 19 March 2024
Netflix unveils a sneak peek at the original film "Joy," chronicling the revolutionary story of Louise Joy Brown, the first baby born through in vitro fertilization (IVF). Written by Rachel Mason and Jack Thorne, the film focuses on Jean Purdy, a nurse and embryologist, portrayed by Thomasin McKenzie of "Last Night in Soho" fame. Purdy, along with surgeon Patrick Steptoe, played by Bill Nighy, and scientist Robert Edwards, portrayed by James Norton, formed a groundbreaking trio that paved the way for IVF. The narrative celebrates their relentless determination and the marvels of science as they faced substantial challenges and skepticism to achieve their vision, ultimately making dreams possible for millions. [ Full Article ] Webinar: ETHICAL CONSIDERATIONS AND MANAGEMENT OF CRYOSTORAGE SAMPLES
International IVF Initiative 19 March 2024

3pm EDT/ 7pm UK/ 8pm CET, Tuesday 26th March 2024. [ Full Article ] |